Axoguard Nerve
Connector®
Axoguard Nerve Connector is a semi-translucent
coaptation aid designed for Connector Assisted Repair®
of a transected nerve with a gap up to 5 mm.

Connect with confidence
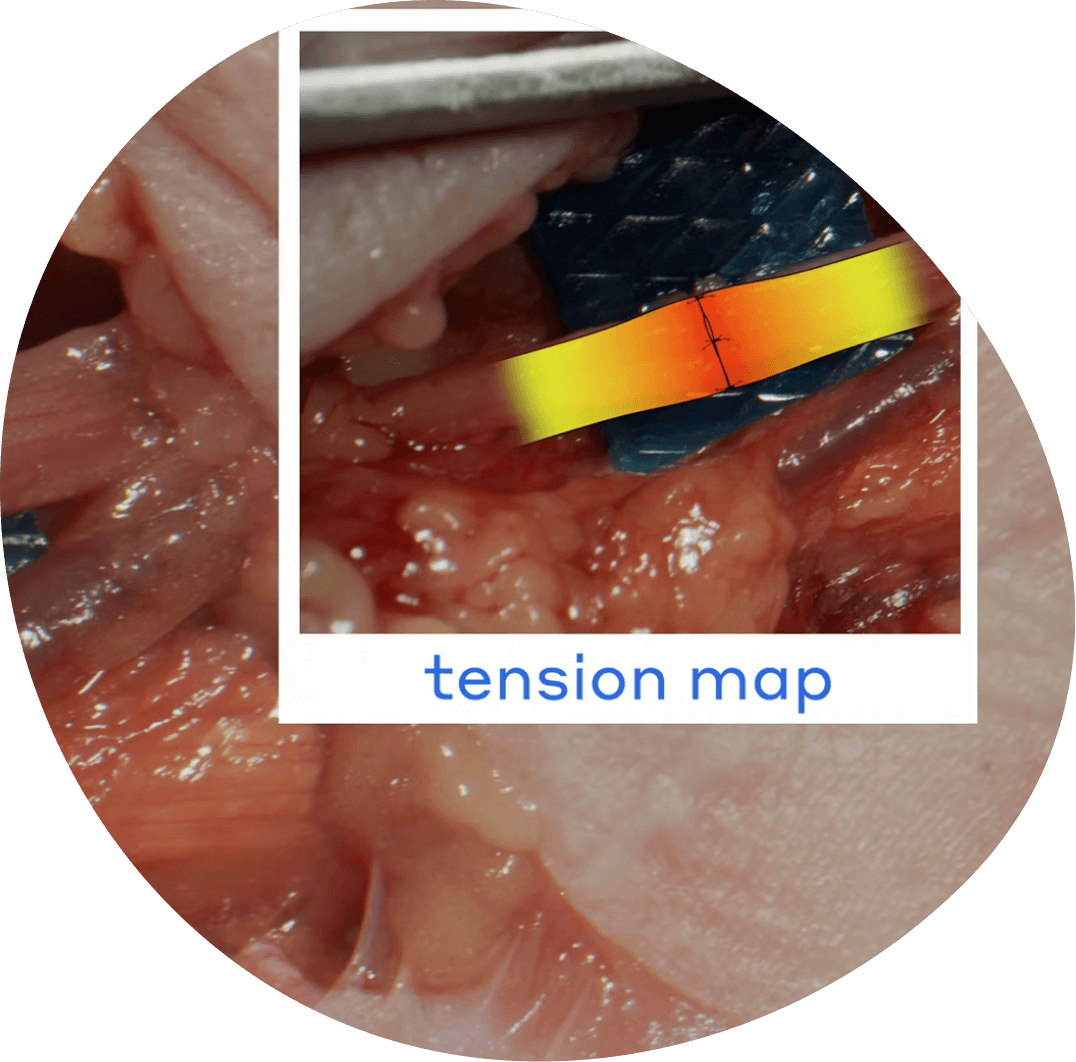
- Axoguard Nerve Connector® is designed to alleviate tension at the repair site and aid in nerve end alignment.
- Porcine small intestine submucosa (SIS) based extracellular matrix allows for vascularization and remodeling into host tissue.1
- Connector Assisted Repair provides an alternative to direct repair that reduces the potential for axonal escape and moves suture-related inflammation away from the critical zone of regeneration.*2
- In a meta-analysis, data showed Connector Assisted Repair led to higher rates of sensory meaningful recovery. Meaningful recovery is defined as MRCC ≥ S3 compared to direct repair alone.3
Ordering and sizing
| Code | Dimensions |
|---|---|
| AGX110 | 1.5 mm x 10 mm |
| AGX210 | 2 mm x 10 mm |
| AGX310 | 3 mm x 10 mm |
| AGX410 | 4 mm x 10 mm |
| AGX510 | 5 mm x 10 mm |
| AGX610 | 6 mm x 10 mm |
| AGX710 | 7 mm x 10 mm |
| Code | Dimensions |
|---|---|
| AGX115 | 1.5 mm x 15 mm |
| AGX215 | 2 mm x 15 mm |
| AGX315 | 3 mm x 15 mm |
| AGX415 | 4 mm x 15 mm |
| AGX515 | 5 mm x 15 mm |
| AGX615 | 6 mm x 15 mm |
| AGX715 | 7 mm x 15 mm |
Understanding Axial Tension: Direct Repair vs Connector Assisted Repair
The importance of biomaterials
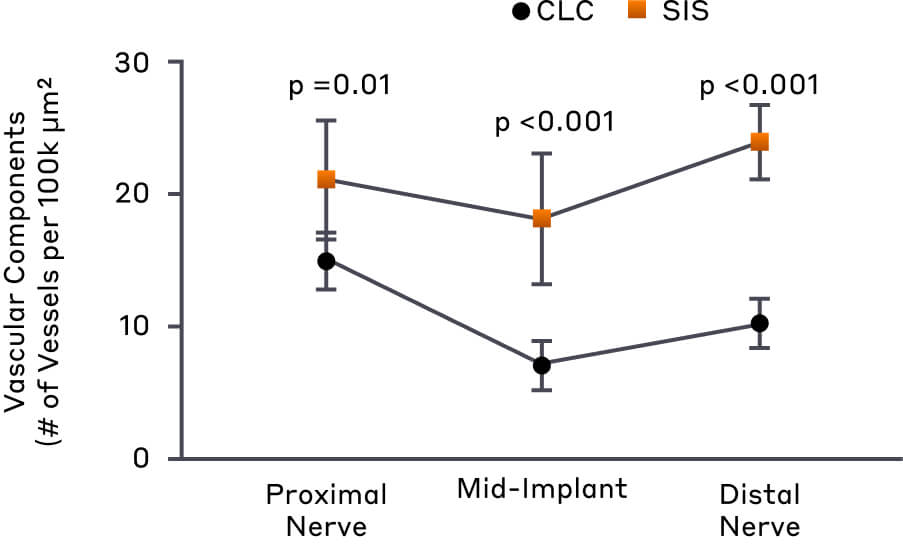
In a pre-clinical animal study designed to characterize host tissue response to different materials following nerve repair, SIS conduits exhibited distinctly different histological profiles compared to a crosslinked collagen product.4
Specifically, the study observed that SIS membranes supported higher vascular component densities and minimal myofibroblast ingrowth into the regenerating nerve cable compared to nerves repaired with a crosslinked collagen product.4
- *
- As seen in a review of 12 clinical studies. Results are limited to the studies reviewed and may not be predictive of all patient outcomes.
- Preclinical animal model results may not be predictive of clinical outcomes.
References:
- Nihsen ES, et al. Bioactivity of small intestinal submucosa and oxidized regenerated cellulose/collagen. Adv Skin Wound Care. Oct 2008;21(10):479-486.
- Ducic I, et al. Refinements of nerve repair with connector-assisted coaptation. Microsurgery. 2017;37(3):256-263.
- Leis A, et al. Comparative effectiveness systematic review and meta-analysis of peripheral nerve repair using direct repair and Connector-Assisted Repair. Plast Reconstr Surg Glob Open. 2024;12(7):e5927. doi:10.1097/GOX.0000000000005927
- Zhukauskas R, et al. Histological comparison of porcine small intestine submucosa and bovine type-I collagen conduit for nerve repair in a rat model. J Hand Surg Glob Online. 2023;5(6):810-817. doi:10.1016/j.jhsg.2023.07.014
IMPORTANT SAFETY INFORMATION
Indications for Use
Axoguard Nerve Connector is indicated for the repair of peripheral nerve discontinuities where gap closure can be achieved by flexion of the extremity. Supplied sterile; single use only.
Contraindications
Do not use in patients with known sensitivity to porcine material.
Not intended for vascular applications.
Potential Complications
Complications associated with nerve repair surgery may include pain, infection, decreased or increased nerve sensitivity, and complications associated with anesthesia.
If inflammation, allergic reaction, or infection occurs and cannot be resolved, careful removal of the device should be considered.
Rx Only
Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
For complete product information, including indications, contraindications, warnings, precautions, and adverse events, please refer to the Instructions for Use (IFU).


