Axoguard
Nerve Cap®
Axoguard Nerve Cap is a porcine small intestine
submucosa (SIS) matrix designed to reduce the
development of symptomatic or painful neuromas.
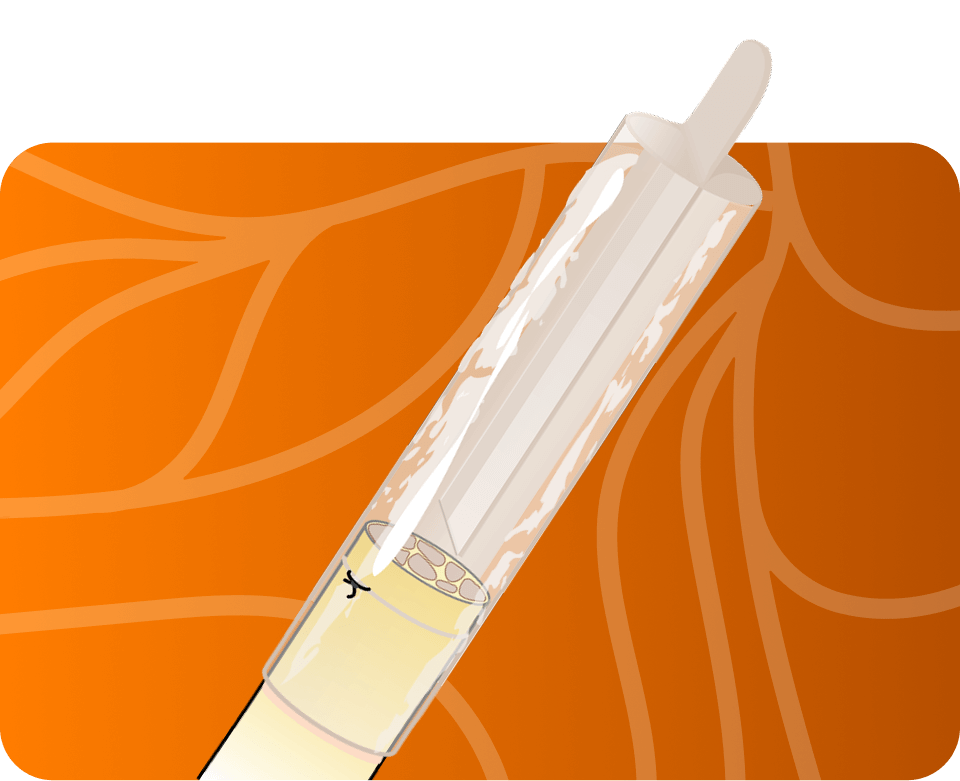
Cap the source of pain
- Axoguard Nerve Cap reduces the development of symptomatic or painful neuromas.
- Proprietary bifurcation designed to minimize pathological axon interaction and help maintain a suitable distance for axonal outgrowth and exhaustion.
- Material gradually remodels to create a physical barrier surrounding the nerve end.
- End tab facilitates burying the nerve end away from mechanical stimulation.
- Suitable for anatomic areas with limited or no musculature.
Ordering and sizing
| Code | Dimensions |
|---|---|
| AGT215 | 2 mm x 15 mm |
| AGT315 | 3 mm x 15 mm |
| AGT415 | 4 mm x 15 mm |
| AGT520 | 5 mm x 20 mm |
| AGT620 | 6 mm x 20 mm |
| AGT720 | 7 mm x 20 mm |
The Axoguard Nerve Cap is intended for single-use only. Any unused portions should be discarded, as specified in the Instructions for Use (IFU). Do not re-sterilize or reuse the device.
See how Axoguard Nerve Cap is used to help reduce the development of painful neuromas.
Watch more videos
This is a promotional video and is not intended to provide clinical guidance beyond the FDA-cleared indications.
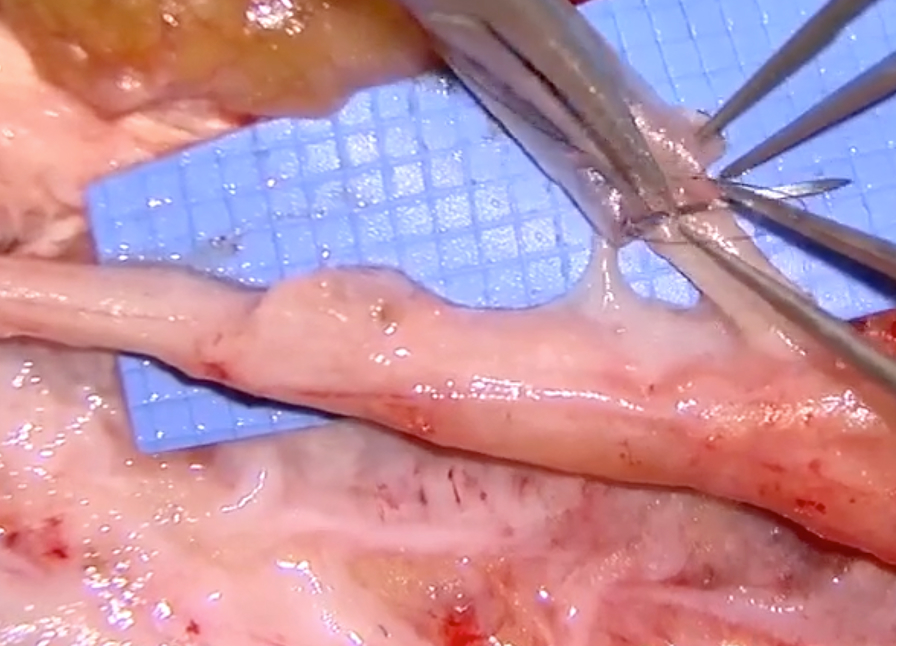
Neuroma Resection with Axoguard Nerve Cap
IMPORTANT SAFETY INFORMATION
Indications for Use
Axoguard Nerve Cap is indicated to protect a peripheral nerve end and to separate the nerve from the surrounding environment to reduce the development of symptomatic or painful neuroma.
Contraindications
Do not use in patients with known sensitivity to porcine-derived materials.
Contraindicated in any patient for whom soft tissue implants are contraindicated, including pathology that would limit blood supply and compromise healing, or evidence of a current infection.
Do not implant directly under the skin.
Not intended for vascular applications.
Potential Complications/Adverse Events
Complications associated with surgery may include pain, infection, decreased or increased nerve sensitivity, and complications associated with anesthesia.
If infection, allergic reaction, or acute/chronic inflammation occurs and cannot be resolved, careful removal of the device should be considered.
Adverse events may include failure to reduce symptomatic neuroma pain, transitory local irritation, infection, allergy, delayed wound healing, and protrusion.
Rx Only
Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
For complete product information, including indications, contraindications, warnings, precautions, and adverse events, please refer to the Instructions for Use (IFU).


