Axoguard Nerve
Protector®
Axoguard Nerve Protector is a porcine small intestine
submucosa (SIS) implant designed to protect injured or
compressed nerves.


Protect what matters
- Axoguard Nerve Protector is designed to be an interface between the nerve and surrounding tissue, minimizing the potential for soft tissue attachments.1
- Porcine SIS-based extracellular matrix allows for vascularization and remodeling into host tissue.1
- Pre-curled design allows for easy handling and circumferential wrapping.
- Hydrates quickly; can be secured using sutures and trimmed to adjust device length.
Ordering and sizing
| Code | Dimensions |
|---|---|
| AG0220 | 2 mm x 20 mm |
| AG0320 | 3.5 mm x 20 mm |
| AG0520 | 5 mm x 20 mm |
| AG0720 | 7 mm x 20 mm |
| AG1020 | 10 mm x 20 mm |
| Code | Dimensions |
|---|---|
| AG0340 | 3.5 mm x 40 mm |
| AG0540 | 5 mm x 40 mm |
| AG0740 | 7 mm x 40 mm |
| AG1040 | 10 mm x 40 mm |
See how Axoguard Nerve Protector is used to wrap nerves after compression or traumatic injuries.

The importance of biomaterials
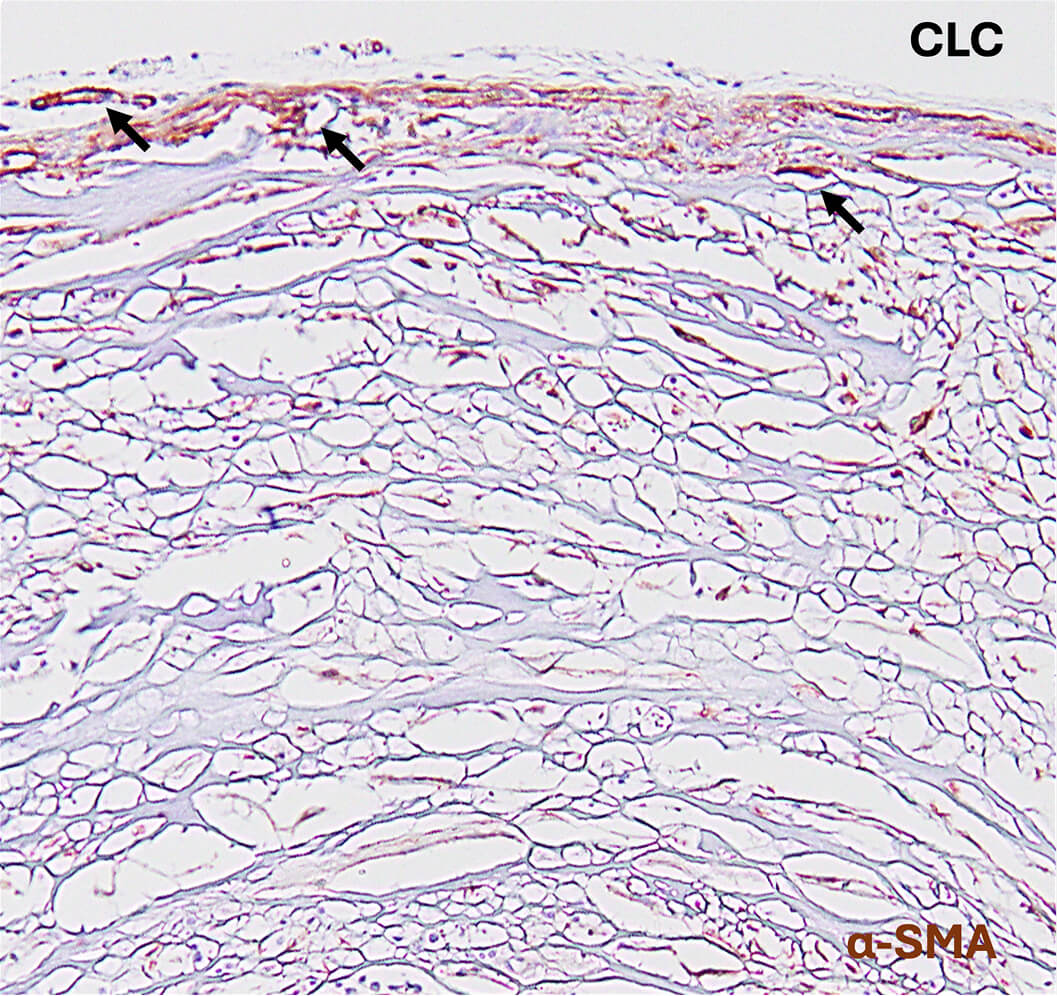
In pre-clinical studies conducted to evaluate host tissue response, the following were observed compared to a cross-linked collagen product:
- SIS membranes supported higher vascular component densities.2
- SIS membranes combined strength, pliability and semi-translucency with greater native cellular ingrowth and vascularization.3
- SIS membranes supported a more favorable tissue environment for nerve regeneration.2



Watch more videos

The Importance of Nerve Protection with Dr. Michael Garcia*

Three Types of Candidates for Nerve Protection with Dr. Sunishka Wimalawansa*

Addressing the Challenge of Treating Non-Transected Nerves with Dr. Meredith Osterman*
- Preclinical animal model results may not be predictive of clinical outcomes.
References:
- Kokkalis ZT, et al. Assessment of processed porcine extracellular matrix as a protective barrier in a rabbit nerve wrap model. J Reconstr Microsurg. Jan 2011;27(1):19-28.
- Zhukauskas R, et al. Histological comparison of porcine small intestine submucosa and bovine type-I collagen conduit for nerve repair in a rat model. J Hand Surg Glob Online. 2023;5(6):810-817. doi: 10.1016/j.jhsg.2023.07.014.
- Zhukauskas R, et al. A comparative study of porcine small intestine submucosa and cross-linked bovine type I collagen as a nerve conduit. J Hand Surg Glob Online. 2021;3(5):282-288. doi:10.1016/j.jhsg.2021.06.006
IMPORTANT SAFETY INFORMATION
Indications for Use
Axoguard Nerve Protector is indicated for the repair of peripheral nerve injuries where there is no gap. Supplied sterile; single use only.
Contraindications
Do not use in patients with known sensitivity to porcine material.
Not intended for vascular applications.
Potential Complications
Complications associated with nerve repair surgery may include pain, infection, decreased or increased nerve sensitivity, and complications associated with anesthesia.
If inflammation, allergic reaction, or infection occurs and cannot be resolved, careful removal of the device should be considered.
Rx Only
Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
For complete product information, including indications, contraindications, warnings, precautions, and adverse events, please refer to the Instructions for Use (IFU).


